The Complete Guide to Electrolyte Analyzers: Importance, Repair, and the Engineer’s Role
In the fast-paced world of a clinical laboratory, few machines are as critical yet as sensitive as the Electrolyte Analyzer. Whether sitting in a core lab or at the patient’s bedside in the ICU, these devices provide the immediate data doctors need to make life-saving decisions.
As a Biomedical Engineer working in the healthcare sector since 2017, I have commissioned, calibrated, and repaired hundreds of these units. From high-throughput clinical chemistry modules to small point-of-care devices, I’ve seen firsthand how a single millivolt of electrical noise can throw off a patient’s diagnosis.
In this comprehensive guide, we will dive deep into why electrolyte testing is vital, how Ion-Selective Electrode (ISE) technology works, how to troubleshoot common failures, and why the Biomedical Engineer is the ultimate guardian of these machines.
1. Why are Electrolytes So Important in Healthcare?
Before we pick up a screwdriver, we must understand the medical importance of what we are measuring. Electrolytes are minerals in the blood and other body fluids that carry an electric charge. They affect how your body functions in many ways, including the amount of water in your body, the acidity of your blood (pH), and your muscle function.
The primary electrolytes measured are:
-
Sodium (Na+): Regulates blood pressure and fluid balance. Abnormal levels can lead to confusion, seizures, or coma.
-
Potassium (K+): Vital for heart function and muscle contraction. Even a slight elevation (hyperkalemia) can trigger fatal cardiac arrest.
-
Chloride (Cl-): Helps maintain osmotic pressure and acid-base balance.
-
Ionized Calcium (iCa{++}): Critical for bone health, blood clotting, and neural transmission.
When a patient crashes in the emergency room, doctors cannot wait hours for a full metabolic panel. They need a quick electrolyte reading. This is why a functional, accurate analyzer is non-negotiable in any modern hospital.
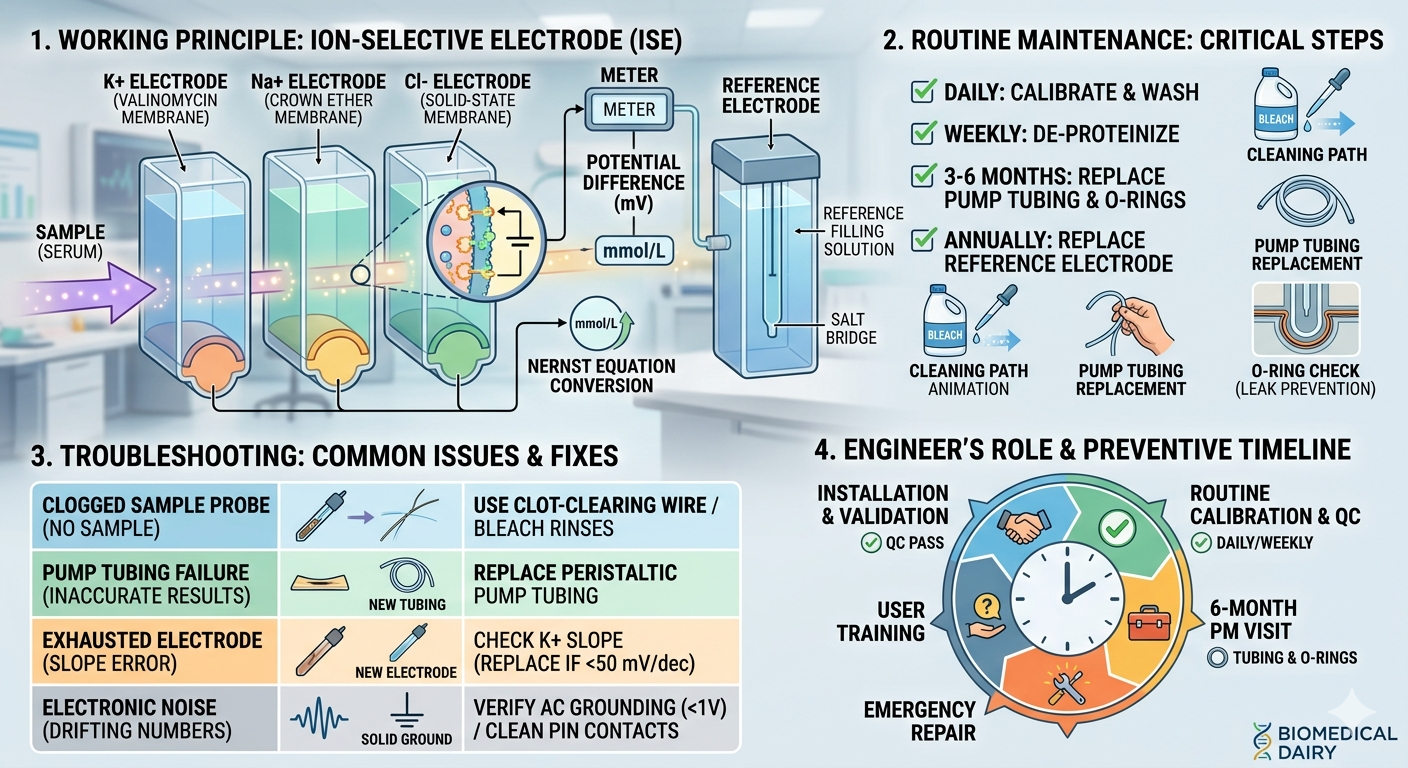
2. Understanding the Technology: How Ion-Selective Electrodes (ISE) Work
To service these machines effectively, an engineer must understand the science behind them. Almost all modern electrolyte analyzers use Ion-Selective Electrodes (ISE).
The technology is based on Potentiometry (the measurement of electrical potential).
The Science in Simple Terms:
Imagine a tube divided by a special membrane. On one side is a known “Internal Filling Solution,” and on the other side is the patient’s blood serum.
-
The membrane is designed to only let one type of ion (like K+) react with it.
-
When the blood sample passes by the membrane, a chemical reaction occurs, generating a tiny electrical voltage (measured in millivolts, or mV).
-
The analyzer compares this voltage to a steady Reference Electrode.
-
The difference in voltage is calculated using the Nernst Equation, converting electricity into a readable concentration (mmol/L).
As an engineer, you are not just fixing a machine; you are maintaining a delicate electro-chemical ecosystem.
3. The Role of the Biomedical Engineer: Beyond Just Repair
As a Senior Service Engineer, I always tell my juniors that our job isn’t just about fixing a broken machine. It is about Preventive Care and User Education.
Machine Validation and Installation
When a hospital buys a new electrolyte analyzer, the engineer must validate it. We run accuracy, precision, and linearity tests using standardized quality control (QC) materials. If the machine cannot reproduce the same result ten times in a row, it cannot go live.
Routine Calibration
Analyzers are programmed to calibrate themselves automatically using “Standard A” (Low concentration) and “Standard B” (High concentration) reagents. However, when the automated calibration fails, the engineer must step in to find out why.
Bridging the Gap Between Engineering and Medicine
Often, a machine is working perfectly, but the technician is getting bad results. This is where the engineer acts as a detective. Was the blood sample hemolyzed (red blood cells ruptured)? Did the technician leave the tube open for too long? Solving a medical equipment problem often requires understanding the pre-analytical lab workflow.
4. Troubleshooting and Repair: Common Problems & Solutions
Over the years, I have categorized electrolyte analyzer failures into three main buckets: Fluidic Clogs, Electrode Exhaustion, and Electronic Noise.
A. The Nightmare of Fluidic Blockages
Because these machines aspirate human blood and serum, proteins and fibrin naturally build up inside the tiny capillary tubes.
-
The Symptom: “Sample not detected,” “Aspiration failed,” or fluctuating readings.
-
The Cause: Fibrin clots or crystallized salt from reagents blocking the tubing.
-
The Engineer’s Fix: I run a heavy de-proteinizing cycle using an enzymatic cleaner or a mild sodium hypochlorite (bleach) solution. If the clog is physical, I use a very fine acupuncture wire to gently clear the sample probe. Warning: Never push a wire too hard, or you will puncture the internal tubing!
B. Worn-Out Peristaltic Pump Tubing
Electrolyte analyzers use peristaltic pumps to squeeze reagents through the system.
-
The Symptom: Inaccurate Na+ or K+ readings.
-
The Cause: Over time, the silicone tubing loses its elasticity and becomes “flat.” If it doesn’t squeeze properly, it doesn’t pull the exact amount of liquid required.
-
The Engineer’s Fix: Replace the pump tubing immediately. In my routine maintenance, I replace these tubes every three to six months, regardless of whether they “look” bad.
C. Electrode “Slope” Failures
Electrodes are consumables—they do not last forever.
-
The Symptom: “Slope Out of Range” error during calibration.
-
The Cause: The membrane is depleted, or the internal filling solution has dried up.
-
The Engineer’s Fix: Check the millivolt reading. If the potassium slope is below 50 mV/dec (normal is usually 54-62 mV/dec), the electrode is dead. Before you throw it away, however, check the Reference Electrode. If the reference junction is blocked, all parameters will fail at once.
D. The Mystery of Grounding and Electronic Drift
Because ISE measures tiny millivolt signals, static electricity or poor grounding can destroy accuracy.
-
The Symptom: Drifting numbers that never stabilize.
-
The Cause: Corrosion on the gold contact pins of the electrode, or a loose electrical ground in the hospital wall socket.
-
The Engineer’s Fix: Clean the contact pins with pure isopropyl alcohol. Ensure the analyzer is plugged into a dedicated online UPS with a solid grounding voltage (neutral-to-ground should be less than 1V).
5. The Engineer’s Preventive Maintenance (PM) Checklist
To keep a clinical lab running without downtime, a strict maintenance schedule is required. Here is the framework I use on the field:
Daily Maintenance (By Lab Users)
-
Standard Two-Point Calibration.
-
Daily wash with de-proteinizing solution.
-
Emptying the waste bottle (biohazard safety!).
Quarterly Maintenance (By the Biomedical Engineer)
-
Visual Inspection: Look for salt leaks at tube joints.
-
Tubing Replacement: Replace peristaltic pump tubes.
-
O-Ring Lubrication: Check the rubber O-rings in the electrode chamber. A tiny air leak will ruin your fluidics.
-
Thermal Check: Some ISE modules require incubation at exactly 37°C. Verify this with a digital thermometer.
Annual Overhaul
-
Replace the Reference Electrode.
-
Replace internal filters.
-
Perform a deep system rinse and electronic check-up.
-
Update analyzer software if the manufacturer has released patches.
Conclusion
The Electrolyte Analyzer may seem like a small black box in the corner of a laboratory, but it is a powerhouse of diagnostic medicine. Maintaining these systems requires a unique blend of chemistry knowledge, mechanical fluidics troubleshooting, and electronic precision.
As a Biomedical Engineer, keeping these machines operational is my contribution to healthcare. It ensures that when a doctor reads a lab report, they are looking at true data, not machine errors.